I am sure that you have heard about chromosomes, but do you know what are they? Most of the time, the DNA (the instruction manual of an individual) of the cell is wrapped and intermingled as if it was a ball of wool or a spaghetti dish, forming a dense net into the cell nucleus (Figure 1, Prophase). However, when the cell starts the process of cell division, chromosomes condense generating the typical X-shaped structures that we are more familiar with (Figure 1, Prophase). Why cells have to form such condensed molecules? It is the way that DNA has of grouping and protecting itself so no part of the genome gets lost in such an important moment such as cell division, in which one cell has to generate two identical daughter cells as we can see in Figure 1.
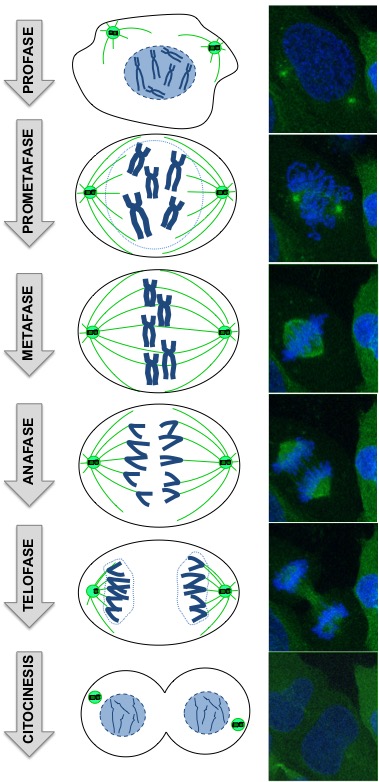
Figure 1. Diagram of the different phases of cell division (left) and microscopic images of each phase captured by real time high-resolution microscopy (right, by M. Alba Abad). The DNA is in blue and the network of microtubules in green, the machinery that helps the chromosomes to be distributed equally into the two identical daughter cells.
Eventual errors in the distribution of the genetic material between the two daughter cells, phenomena known as aneuploidy, give rise to alterations of the number of chromosomes, which are the cause of many types of cancer and different congenital diseases, such as Down’s syndrome (caused by an error in the chromosome 21 distribution during cell division). For this reason, it is very important to understand all the mechanisms and processes needed to allow a correct cell division.
To ensure an equal distribution of genetic material and that every chromosome arm goes to a daughter cell, the centromeric region plays a fundamental role (Figure 2). The centromere acts as a hinge binding the two chromosome arms, like the two parts of scissors or pliers, and it is the part from where chromosomes are separated. The centromere characteristics at a molecular and cellular level are extremely complex, both for its structure and for the mechanisms that control its function, which depend on many different cell factors.
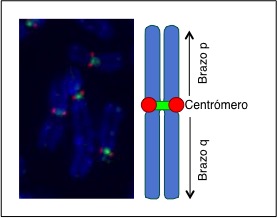
Figure 2. Micrograph of condensed human chromosomes (left) and diagram of a chromosome (right). We can distinguish the DNA (blue) and the centrosome of every single chromosome (red and green).
My main interest during the last four years has been the study of the centromere by using human artificial chromosomes (HACs) (Figure 3). HACs are tiny synthetic chromosomes generated in the laboratory, which have an autonomous replication in the cell and show a similar stability as the natural chromosomes during the cell division. Since the description of the first HAC in 1997, different laboratories throughout the world, including ours, have generated a big number of them through diverse genetic engineering technologies. Since their first description, HACs have been used in basic research in cell and molecular biology, usually for the study of chromosome structure and function, as these chromosomes are not essential for the cell viability. They have also been used for gene regulation studies, as HACs can integrate very big DNA fragments containing entire genes together with their regulatory sequences. Due to these last characteristics, HACs have been suggested to have a big potential for being used in gene therapy, thus bypassing the use of viruses to deliver healthy genes to the patient cells with genetic abnormalities to prevent immunogenic responses as a consequence of the viral infection.
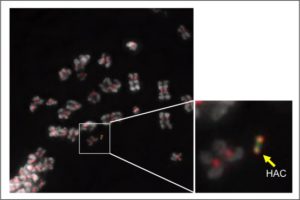
Figure 3. Micrograph of different chromosomes in a cell containing a HAC (magnification on the right). Centromeres are labelled in red and the chromosomes in grey.
Thanks to the use of HACs, scientists have been able to study the minimal requirements to assemble and maintain such complex machinery as the centromeres that are necessary, as stated above, to ensure proper chromosome segregation during cell division. Contrary to what has been thought in the past, our group has recently shown that centromeres are not regions of “silenced” DNA, but they contain factors and modifications that are typical of active genes. Indeed, the removal of these particular modifications, typical of active genes, disrupt the structure and function of the centromere, thus impairing its proper role in cell division.
Although there is a lot to know about the function and maintenance of the centromeres, HACs have proven a unique tool for the study of these chromosomic regions and for the study of the mechanism of chromosome segregation during cell division. Beyond their use in basic research, it is very likely that in the future we hear news about the HACs as useful tools for other applications. Time will tell.
By Dr Óscar Molina Campoy, Postdoctoral researcher, Wellcome Trust Centre for Cell Biology, University of Edimburgh. SRUK Constituency of Scotland.
More info: