Pregnancy, a unique and special time in women’s life. Elisabeth Hasselbeck described pregnancy as “the most beautiful and significant life-altering event with motherhood in her experience” and Beyoncé commented that “the most powerful creation is to have life growing inside of you. There is no bigger gift”. Indeed, pregnancy is a miraculous event – but, what many people do not realise, is that it is also biologically highly demanding. Like a ballerina who makes difficult choreographies look easy as she is gliding on the stage, the shine of pregnancy hides the intricate changes the women’s body undertakes to successfully support both the development of the embryo and the mother.
Due to its complexity, pregnancy can lead to a variety of complications with a range of severities; such as pre-maturity, gestational diabetes or cardiovascular complications. The latter are the leading causes of pregnancy complications in Western countries. Around 10-15% of pregnancies involve hypertensive disorders, which refer to the elevated maternal blood pressure (gestational hypertension) that can be associated with protein secretion in urine (known as preeclampsia) or worse symptoms, such as seizures (eclampsia) or liver failure (HELLP: haemolysis, elevated liver enzymes, and low platelet count) [1,2,3,4,5,6]. Although maternal mortality due to hypertensive disorders of pregnancy has significantly been reduced, this condition can still lead to very high morbidity during pregnancy and postnatally in the mother (e.g., multiple organ failure and placental abruption) and in the foetus (e.g., intrauterine growth retardation or death) [2,3,4,7]. In addition, maternal complications, as well as other adverse stimuli at this life stage, can program the mother and offspring to suffer non-communicable diseases later in life, such as cardiovascular abnormalities (e.g., hypertension or stroke), diabetes and subfertility [4,8,9,10]. Currently, in clinical practice, women’s medical history prior to the pregnancy and measurements from perinatal visits are used to predict and prevent or lessen these complications.
But… is this enough as a preventive tool?
We believe that, aside from certain prior diseases, which are associated with pregnancy complications, prenatal suboptimal health might also predict the potential to develop these complications during pregnancy when the body is under stress. In addition, we also believe that suboptimal adaptation to these stressors during pregnancy is enough to tax the system and program for non-communicable diseases later in life, even if there are no obvious symptoms during pregnancy.
To investigate the role of suboptimal health and adaptation on pregnancy complications and programming for non-communicable diseases, we first need to understand what is considered a “normal” pregnancy. In other words: what different patterns do women have during pregnancy, and which ones lead to complications and maladaptation?
It is well established that pregnancy is associated with a range of expected changes in both the maternal psychology and physiology, such as:

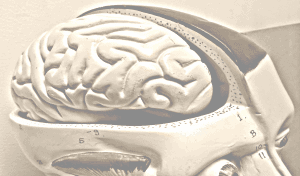
Figure 1. Illustration of the changes undergone by the women’s body during pregnancy. Image modified from publicly available images at Pixaby.
- Physical changes: increase in body weight and shape.
- Hormonal changes: the stress hormone, cortisol, increases and, together with an increased insulin response, allows the body to use fat for maternal energy. In this way, glucose and amino acids can be preserved to provide energy for the foetus [11,12].
- Respiratory changes: by the third trimester, the mother’s energy requirements increase to around 200 kcal per day to fulfil the metabolic demands. In addition, her lungs adapt to increase oxygen intake by approximately 20% [11].
- Cardiovascular changes: circulating blood volume needs to increase to allow supply for both individuals. To achieve this, her heart adapts leading to an increase of the maternal cardiac output by 40% in the third trimester. This is associated with lower blood pressure in the first two trimesters, but higher blood pressure in the third. Importantly, increased blood volume requires the body to produce more blood cells, which demands higher levels of iron, an essential metabolite in this process [12].
These changes are the tip of the iceberg and many unknowns remain to be discovered. For instance, how does the maternal heart shape change throughout pregnancy? Furthermore, there is an expectation that, in the absence of diseases, all women undergo these changes during pregnancy in a similar way, and that the maternal body returns to preconception physiology following delivery of the baby. But, is this true? For example, does the maternal heart return to its initial shape?
We have joined forces with the Cardiovascular and Women’s health departments at the University of Oxford to shed light on these uncertainties with the OXWATCH (Oxfordshire Women’s and Their Children’s Health) study. As part of this project, 250 women between 18 and 40 years old that were planning a pregnancy were followed from pre-conception to post-pregnancy. During these follow-ups, an extensive amount of information was collected via genetic, biophysical, socioeconomic, behavioural and psychological assessments. This information has allowed us to start addressing some of the uncertainties by tackling the following questions:
How does fat distribution change through normal pregnancy? Does it recover? And then, how does this relate to cardiovascular health?
How does heart and vessel physiology change throughout “normal” pregnancy and recovers in relation to prior health status?
Do women who suffer from anxiety and depression through pregnancy and postnatally have a different psychological threshold pre-conception?
Disease is often a triad of biological, psychological and environmental factors. We hope this work starts to unravel the patterns of pregnancy in a non-disease state, and that it also contributes to the discovery of suboptimal recoveries after pregnancy that, in combination with environmental stressors, can lead to non-communicable diseases later in life.
* * *
By Helena Rodríguez-Caro (@HelenaRodriCaro) and Annabelle Frost. Helena is a post-doctoral researcher at the Department of Physiology, Anatomy, and Genetics (University of Oxford) and Annabel is a post-doctoral researcher at the Cardiovascular Clinical Research Facility (University of Oxford).
More information:
- Khan KS et al. (2006).
- Regitz-Zagrosek V et al. (2018).
- Web resource: “Saving lives, improving mother’s care”. Slides by Knight M BK.
- Fox R et al. (2019).
- James PR et al. (2004).
- European Society of Gynecology (2011).
- Rezk M et al. (2015).
- Staff AC et al. (2016).
- Rodriguez-Caro H & Williams SA (2018).
- Arnott C et al. (2019).
- Abduljalil K et al. (2012).
- Soma-Pillay P et al. (2016).