The release of ‘The Origin of Species’ by the famous naturalist Charles Darwin in 1859 caused a paradigmatic change in the natural sciences. In his book, Darwin proposed that species (including Homo sapiens) were not created by an omnipotent God, but were the result of the evolutionary process, whose main driving force is natural selection. Darwin’s work was initially mocked, but with time it was finally accepted by the scientific community. In the early 20th century, the theory of evolution was fused with the genetic theory by Mendel, which formed the basis of what we know today as modern synthesis or Neo-Darwinism. Neo-Darwinism establishes that the random mutation of genes, generation after generation, is what generates the evolutionary novelty that justifies the wide variety of species that live on our planet.
For a long time, biology students have learned the neo-darwinian thesis during their degrees. However, not all of them know that there are scientists that disagree with the predominant idea that establishes that the accumulation of random mutations and their inheritance throughout generations as the driving force for the origin of new species. Among the defenders of alternative theories was one of the most important scientists of the 20th century: Lynn Margulis (1938-2011). Margulis, who studied biology, dedicated her academic career to the study of evolution. Unlike most of the evolutionists at the time (which were mainly zoologists), she thought that the basis for the study of evolution was the cell, which is the elementary unit of life. Therefore, she focused her research on microorganisms, especially in bacteria, since she considered them the origin of the current biological diversity. By the end of the 60s, she introduced the Endosymbiotic Theory to the rest of the scientific community.

The Endosymbiotic Theory argues that the eukaryotic cells (whose DNA is inside the cell nucleus) have a bacterial origin. Margulis gives a series of reasons that explain this event. Roughly 250 genes of the 30,000 contained in the human cell nucleus share identical sequence with bacterial genes. She also points out that the DNA within mitochondria and chloroplasts (components of animal and plant cells, respectively) are very similar to bacterial DNA. This suggests that these cellular components were once independent in the past, and somehow integrated themselves in a different bacteria, and subsequently generated the first eukaryotic cell. Margulis labelled this type of association as symbiotic fusion.
In biology, the term symbiosis refers to the interaction between organisms of different species, which both obtain mutual benefits from their relationship. The prime example is the association of a fungus with a cyanobacteria, which come together to form a lichen. Lichens cannot be classified as fungi or cyanobacteria, even if they are made out of them. The individual organisms are so tightly connected to each other that they are unrecognizable on their own, and they could not survive without the other. This is why lichens are considered a result of symbiogenesis; their interaction is so strong that both fungus and cyanobacteria lose their identity and form a completely new species.
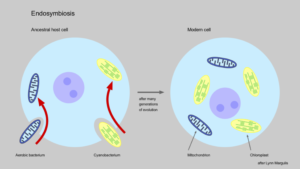
According to Margulis, symbiogenesis is what developed the variety of species that exist and have ever existed. In her book ‘Acquiring genomes’, written together with Dorion Sagan in 2002, Margulis gives plenty of examples of new species that emerged as the result of symbiogenesis. Nevertheless, the main criticism from neo-darwinian scientists against the theories of Margulis is that the amount of newly arisen species via this mechanism is a minority. They claim that the huge majority of genetic variation originates from the accumulation of random mutations in the DNA. Lynn Margulis argues that this mechanism is not likely to drive speciation, since random mutation generally leads to the loss of functionality in genes, and finally to disease. In contrast, she proposes that the source of evolutive novelty is the acquisition of entire genomes from some organisms to others, a feature of symbiogenesis. Moreover, this explanation gives some insights into the mystery of the fossil record. The fossil record shows dramatic changes in the morphology of species; these extreme changes may suggest that species do not evolve gradually, but in an abrupt way.
A nice example of symbiogenesis is the relationship between the squid Euprymna scolopes and the bacteria Vibrio fischeri. The squid has a set of genes that controls the growth of a special tissue that helps it to capture a single strain of V. fischeri. Once the bacteria are inside the squid, the tissue degrades. However, if by any chance the animal loses its symbiotic partner, the tissue is able to regenerate, so that it would capture the bacteria again. In this association, V. fischeri receives nourishment from the squid, whereas the squid benefits from the bioluminescence generated by the bacteria. Bioluminescence dazes the potential predators of the squid, since they confuse the light with the reflection of the moon in the water. This is a clear example of how symbiogenetic relationships are genetically regulated.
Although the scientific community is still debating the true origin of life, the theory of symbiogenesis as the vital driving force of speciation is one of the most important contributions to evolutive biology since the times of Darwin. The new discoveries of modern genetics and molecular biology support the Symbiogenetic Theory; through the comparison of genomes from many species, we could track the footprints of symbiogenetic events throughout evolution. The new fields of scientific knowledge could be hiding the answer to the mystery of evolution. Could symbiogenesis be an alternative story of the origin of life?
References:
https://elpais.com/elpais/2015/08/19/ciencia/1440001134_537219.html
https://es.wikipedia.org/wiki/Cyanobacteria
By Ignacio Jiménez, PhD student, Wellcome Trust Centre for Cell Biology, University of Edinburgh. Scotland SRUK constituency.




