Neurons are my favorite cells – I’m a neuroscientist for this reason! – and although it is true that our brain is not only made up of neurons (astrocytes, microglia and oligodendrocyte are also very important cells in our brain), neurons are mysterious and still keep many secrets about their function that make them very special.
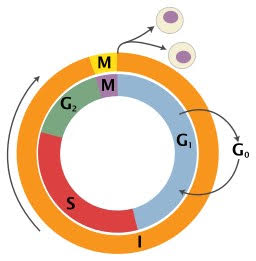
Figure 1. The cell cycle and its phases. During the G1, S (synthesis) and G2 phases, all the cellular components that are going to be part of the daughter cells duplicate, in addition to verifying that there are no errors during this duplication. During mitosis (M) cell division occurs resulting in the two daughter cells. Phases G1, S and G2 together are also referred to as Interphase (I). Mature neurons maintain their cell cycle paused in the G0 phase but they reactivate it and proceed to G1 when DNA damage repair is needed. Photo credit: Richard Wheeler (Zephyris), 2006, available at Wikipedia.
The neuronal cell cycle is one of the functions that interest me the most when talking about neurons. The cell cycle is the mechanism that eukaryotic cells (that is, cells with genetic material confined to a defined nucleus) use to equally divide their genetic material, or DNA, between two daughter cells. The cell cycle is like a clock that marks several phases during which the cell prepares to divide (see Figure 1). As in a chain reaction, the cell activates one phase after another; specialized machinery is activated in each phase in order to prepare for cell division and to monitor that the cell cycle occurs efficiently and accurately, thus avoiding errors that could lead the daughter cells to catastrophe.
All the cells in our body have the specialized machinery needed to activate the cell cycle and are able to generate new cells to replace those that have died for different reasons. But neurons are not like other cells and even though they also contain the cell cycle machinery, they don’t necessarily use it for cell division. This occurs because once neurons mature or differentiate into adult neurons, they stay in the G0 phase (inactive phase) of the cell cycle and lose the ability to form daughter cells (although there are some areas in the adult brain where neurogenesis, or formation of new neurons, does occur, but under very specific conditions). If mature neurons don’t divide, what happens to all the machinery of the cell cycle? Is it also lost? The answer is “no”, and here’s why.
A few years ago, a research group observed that mature neurons can exit the G0 phase and reactivate their cell cycle, but not to form daughter cells [1]. It turns out that neurons reactivate their cell cycle in response to direct damage to their DNA. The DNA molecule is made up of two chains that hold our genetic code inside each cell of our body, and it is very fragile. Certain damaging conditions, such as radiation or oxidative stress, can cause DNA damage in the form of breaks in one or both of its strands. Neurons, as well as other cell types, can repair these DNA breaks (see Figure 2), but to do so, they must first detect them. This process occurs through cellular detection programs that flag DNA lesions in a similar way that traffic cones warn motorists about a bump on the road. In neurons, these DNA damage detection and repair programs cannot occur without first exiting the G0 phase and reactivating their cell cycle. Once DNA break is located and repaired, neurons return to their normal state in the G0 phase of the cell cycle.
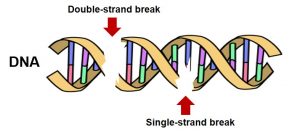
Figure 2. DNA can suffer different types of injuries, but cuts in one or both strands (single and double-strand breaks) are the most problematic ones. Photo credit: Wikimedia.
This discovery is very important because it shows us how important the neuronal cell cycle machinery is to protect neurons from direct damage to their DNA despite not being used to produce daughter cells. This biological scenario triggers questions such as what would happen if cell cycle reactivation in neurons occurred abnormally or if neurons did not return to G0 after the damage was repaired but instead advanced to the mitosis phase (that is, when daughter cells are formed).
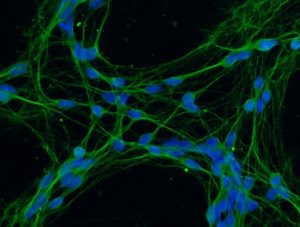
Figure 3. Mature human neurons grown in the laboratory. The nuclei of neurons, where DNA is found, are seen in blue, while the bodies of neurons are seen in green. Photo credit: Irina Vázquez Villaseñor.
These questions and many more are being investigated under normal conditions and in relation to the development of neurodegenerative diseases, such as Alzheimer’s disease. Now, scientists wonder if one of the causes of brain degeneration observed in these diseases could be due to an abnormal start of the cell cycle in mature neurons in response to chronic DNA damage and whether this could be a target for treatment. Even though we still lack many clues that can help us solve the mystery of the neuronal cell cycle and its role in the neuronal DNA damage response, we are ready and excited for what we can discover in the future about these extraordinary cells.
* * *
By Irina Vázquez Villaseñor (@Irina_Grishin). Postdoctoral research at the Sheffield Institute for Translational Neuroscience, University of Sheffield, UK.
More information: